For hydrogen, there is only one electron and so the nuclear charge ( Z) and the effective nuclear charge ( Z eff) are equal. This is the pull exerted on a specific electron by the nucleus, taking into account any electron–electron repulsions. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.\). 
Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). For the main group elements (groups designated with a. The number of valence electrons in one atom of each element is easily determined based on its position in the periodic table. 
These variables allowed Mendeleev to place each element in a certain row (called a period) and column (called a group). Thus, Na in Group IA has 1 valence electron, whereas C, in Group IVA has 4 valence electrons. The periodic table is arranged by atomic weight and valence electrons. Conveniently, the number of valence electrons for the A Group elements is equal to the group number. The elements in a group share the same number of valence electrons and thus have many common chemical and physical properties. This is determined based on the number of electrons that would be added, lost, or shared if it reacts with other atoms. One last trend that occurs on the periodic table is the trend of the number of valence electrons. Many periodic tables list numbers for element groups, which are columns of the periodic table. Valence describes how easily an atom or radical can combine with other chemical species. Free Gift for you: Interactive Periodic Table Valence electrons are the electrons that reside in the outermost energy level of an atom and are, therefore, the most accessible for the formation of chemical bonds. The words valence and valency have two related meanings in chemistry. In this way, the elements of the same group show similar chemical properties and they also have the same number of valence electrons. They are soft and can be cut easily with a kitchen knife.Īlso all the elements of group 1 have one valence electron.Īll the elements of group 18 are chemically inert (that means they do not easily react with other elements).Īnd all the elements of group 18 have a complete octet (that means they have 8 electrons in their outer shell). The s-, p-, and d-block elements of the periodic table are arranged into 18 numbered columns, or groups. The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons.Īll the elements of group 1 are highly reactive to water. The easiest way to find the number of valence electrons is to go by the element group in the valence periodic table. 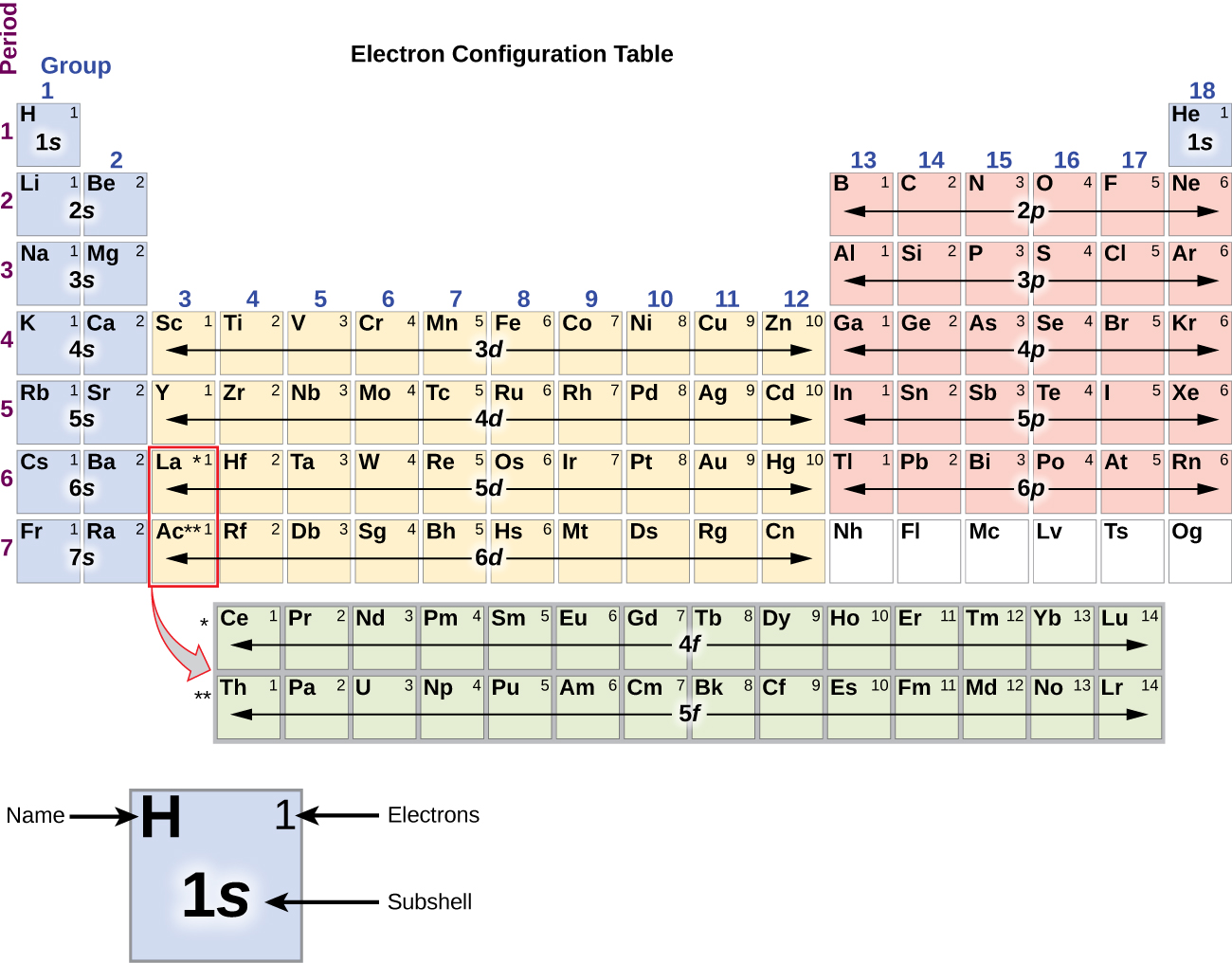
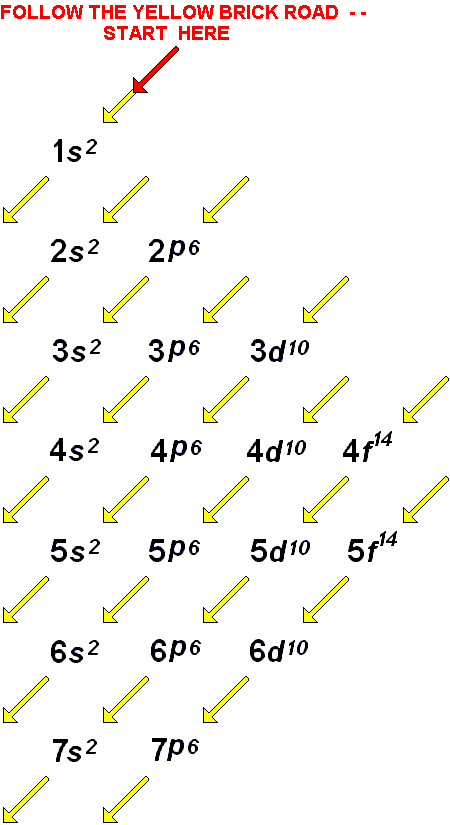
How to Find the Number of Valence Electrons. Thus beryllium has an He s2 electron configuration. Beginning with beryllium, we see that its nearest preceding noble gas is helium and that the principal quantum number of its valence shell is n 2. Learn for free about math, art, computer programming, economics, physics, chemistry, biology, medicine, finance, history, and more. Solution: The group 2 elements are in the s block of the periodic table, and as group 2 elements, they all have two valence electrons. Stuck Review related articles/videos or use a hint. A neutral Ba atom has valence electron (s). So, they can have more than 8 valence electrons. Complete the statement by entering a number. There are total 18 vertical columns on periodic table. Metals in the middle of the periodic table become more stable by emptying a shell, half-filling it, or completely filling it. The table is arranged such that elements that have the same number of valence electrons fall under the same group. Valence electrons are responsible for the reactivity of an element. Explanation: The periodic table indicates the number of valence electrons an element has through its arrangement into groups and periods. Groups are the vertical columns on the periodic table. Atoms in Groups 13 and 18 have 3 and 8 valence electrons, respectively. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
